
How are the elements grouped in the periodic table?
- Group 1 contains elements containing only 1 valence electron in their atoms, these are called alkali metals such as lithium, sodium, potassium, etc. ...
- Group 2 contains alkaline earth metals like beryllium, magnesium, calcium, and so on have 2 valence electrons. ...
- Group 17 contains halogens like fluorine, chlorine, bromine, etc. ...
How many elements can you find on a periodic table?
Unless you currently work in a field that involves science, the days of memorizing the elements of the period table are long behind you. There are currently 118 known elements on the periodic table, and we’ve put 20 of them below to test your smarts.
How do you find elements in the periodic table?
How do you find elements in the periodic table? To find the number of electrons an element has, locate it on the periodic table of elements, find the atomic number, and note the number of protons; because atoms are naturally electrically neutral, the protons and electrons are usually equal. Look at the oxidation number for further information.
How are families of elements arranged in a periodic table?
Three systems have been used to number families and groups:
- The older IUPAC system used Roman numerals together with letters to distinguish between the left (A) and right (B) side of the periodic table.
- The CAS system used letters to differentiate main group (A) and transition (B) elements.
- The modern IUPAC system uses Arabic numbers 1-18, simply numbering the columns of the periodic table from left to right.
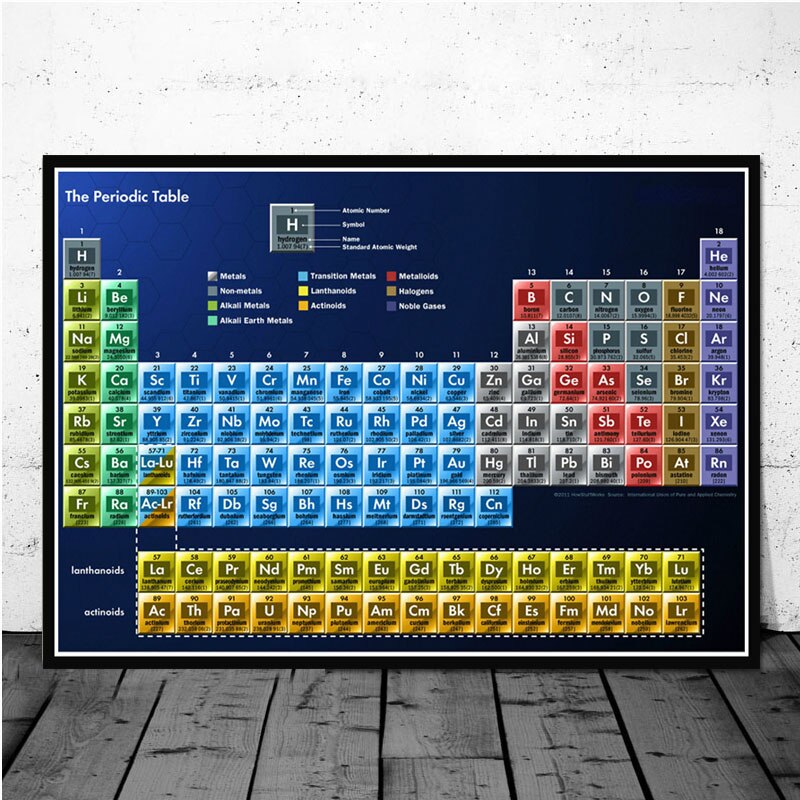
How does the periodic table organize the elements?
The classic Periodic Table organizes the chemical elements according to the number of protons that each has in its atomic nucleus. (Image credit: Karl Tate, Livescience.com contributor)
Which group of the periodic table is alkaline earth metals in?
Alkaline-earth metals: The alkaline-earth metals make up Group 2 of the periodic table, from beryllium (Be) through radium (Ra). Each of these elements has two electrons in its outermost energy level, which makes the alkaline earths reactive enough that they're rarely found alone in nature. But they're not as reactive as the alkali metals. Their chemical reactions typically occur more slowly and produce less heat compared to the alkali metals.
What are the groups of metals that are radioactive?
All are radioactive. The actinides and the lanthanides together form a group called the inner transition metals. Transition metals: Returning to the main body of the table, the remainder of Groups 3 through 12 represent the rest of the transition metals.
What are the elements in the actinides?
Actinides: The actinides line the bottom row of the island and comprise elements 89, actinium (Ac), through 103, lawrencium (Lr). Of these elements, only thorium (Th) and uranium (U) occur naturally on Earth in substantial amounts. All are radioactive. The actinides and the lanthanides together form a group called the inner transition metals.
What is table salt?
The table salt in your kitchen, for example, is a marriage between the alkali metal sodium and the halogen chlorine. Noble gases: Colorless, odorless and almost completely nonreactive, the inert, or noble gases round out the table in Group 18.
How many elements were there at the time of Mendeleev?
There were only about 60 elements known at the time, but Mendeleev realized that when the elements were organized by weight, certain types of elements occurred in regular intervals, or periods. Today, 150 years later, chemists officially recognize 118 elements (after the addition of four newcomers in 2016) and still use Mendeleev's periodic table ...
Why is the period of sodium longer?
Moving down the table, periods are longer because it takes more electrons to fill the larger and more complex outer levels. The columns of the table represent groups, or families, of elements.
How are the columns in the periodic table organized?
The rows and columns are organized by precise characteristics. The elements that are in the same column or in the same rows have common characteristics. For example, magnesium (Mg) and sodium ...
Which two elements are on the top of the periodic table?
The Two Elements At the Top. You are probably wondering about the elements that are alone on the top: hydrogen (H) and helium (He). These two are special elements for different reasons. Hydrogen (H) does not have a single neutron in its neutral form, only one proton and one electron.
Why are valence electrons important?
— Marty Rubin. All the elements in each group have the same number of electrons in their outer orbitals, also known as valence electrons. These electrons are important because they are involved in the chemical bonds with other elements.
How many electrons does helium have?
Helium (He) is unique among all the elements. It only has two electrons in its outer orbital, also known as the valence shell. All the other noble gases (group 18) have eight electrons in their outer orbital or valence shell.
How many orbitals does an element have?
If you look at all the elements on the top row or, in other words, the elements in the first period, you will see that all of them have one atomic orbital for their electrons. Then, the elements on the second row, or second period, are characterized by having two atomic orbitals in their electrons.
How to read valence electrons?
You have to read groups from left to right. All the elements in the first column, or group one, have one valence electrons (one electron in their outer shell). All the elements in the second column, or group two, have two valence electrons. But all the elements in the third group (group three), have thirteen valance electrons. From then on, you have to add an electron for every group until reaching 18. Simply, counting the columns will allow you to know how many electrons each element has on its outer shell. There are a few exceptions to this, though, because some elements are transition elements that add electrons.
Why do all elements have one thing in common?
Because they all have one thing in common: their respective valence shells are full. This is how the periodic table is organized. Understanding that the position of each and every one of the elements is useful in understanding their properties.
How are elements arranged in the periodic table?
All the elements in the table are arranged in rows and columns; the rows runs from left to right and are called periods while the columns run up and down and are called groups. Elements in the same group share similar properties.
Which element is in the gaseous phase?
Samples of four Group 15 elements, antimony, arsenic, bismuth, and phosphorus, are in the gaseous phase. An atom in the ground state of which element …
