
How many identified elements does the periodic table show?
SHOW ANSWER. Periodic table shows 118 identified elements. So, option C is your answer. Hope this helps! Answer from: erikloza12pdidtx. SHOW ANSWER. C. 118. Explanation: Depending on what version you are using, most periodic tables have more than 92 elements on a periodic table.
How many naturally occurring element are in the periodic table?
The naturally occurring elements in the periodic table include the 94 chemical elements from atomic number 1 to 94. Six of these 94 elements (technetium, promethium, astatine, francium, neptunium, and plutonium) are available in extreme trace amounts.
How are elements arranged in the current periodic table?
The modern periodic table
- Video. While you're watching, look out for where different elements are found in the periodic table. ...
- The periodic table. The elements are arranged in order of increasing atomic number. ...
- The arrangement of the periodic table. On the periodic table, metals are found on the left and in the middle, and non-metals are on the right.
- Working scientifically. ...
What are the different element groups in the periodic table?
- Boron (B)
- Aluminum (Al)
- Gallium (Ga)
- Indium (In)
- Thallium (Tl)
- Nihonium (Nh)
See more

Are there 119 elements?
Ununennium, also known as eka-francium or element 119, is the hypothetical chemical element with symbol Uue and atomic number 119....UnunenniumAlternative nameselement 119, eka-franciumUnunennium in the periodic table26 more rows
How many elements exist in total?
This list contains the 118 elements of chemistry. For chemistry students and teachers: The tabular chart on the right is arranged by Atomic number. The first chemical element is Hydrogen and the last is Ununoctium.
How many of the 118 elements in the periodic table are metals?
95Around 95 of the 118 elements in the periodic table are metals (or are likely to be such). The number is inexact as the boundaries between metals, nonmetals, and metalloids fluctuate slightly due to a lack of universally accepted definitions of the categories involved.
What is the name of 118 elements?
118 Elements and Their Symbols and Atomic NumbersName of the ElementSymbol of the ElementAtomic NumberArgonAr18PotassiumK19CalciumCa20ScandiumSc2166 more rows
Is fire an element?
Fire is one of the four classical elements along with Earth, Water and Air in ancient Greek philosophy and science. Fire is considered to be both hot and dry and, according to Plato, is associated with the tetrahedron.
Which is a pure element?
A pure element is one which constitutes of similar kind of atoms. Thus, sodium is a pure element having only sodium atoms. While glass and cement are made up of different kinds of atoms.
What is the longest element name?
Praseodymium is the second-longest name on the periodic table. The prize for the longest element name goes to rutherfordium, named after the famous New Zealand physicist Lord Ernest Rutherford who won a Nobel Prize in chemistry.
What are the 3 types of metals?
How many types of metal are there?Ferrous (contains iron)Nonferrous (contains no iron)Alloys (contains multiple metals and other elements)
How many metals are in the world?
Metals are arranged on the left side of the periodic table and there are five kinds of metals: Alkaline earth metals, Alkali metals, Transition metals, Actinides and Lanthanides. There are a total 92 elements out of which 70 are metals.
Are there 112 or 118 elements?
The Periodic Table is made up of 118 Elements.
What are the main elements?
According to the five elements theory, everything in nature is made up of five elements: Earth, Water, Fire, Air, and Space.
Is water an element?
Water is a compound because of the following reasons: It is composed of two different elements, hydrogen and oxygen which cannot be separated by physical methods. They can be separated only by chemical methods.
Are there 92 or 94 natural elements?
Elements 1 through 92 (except for elements 43 and 61) occur naturally on Earth, although some are only present in extremely small quantities. The elements following uranium on the periodic table are only produced artificially, and are known as the transuranium or transuranic elements.
Are there 94 of 92 naturally occurring elements?
In addition to the 94 naturally occurring elements, several artificial elements have been produced by human nuclear physics technology. As of 2021, these experiments have produced all elements up to atomic number 118.
How many elements are theoretically possible?
137 elementsThere can be no more than 137 elements. This limitation of the number of elements follows from the fact that there is a maximum speed in the Universe. That is, from the limit of the speed of light in a vacuum...
Why are there only 92 naturally occurring elements?
The usual textbook answer is 91. Scientists used to believe that, except for the element technetium, all the elements up to element 92 (uranium) could be found in nature. However, it turns out there are other elements that occur in trace amounts naturally. This brings the number of naturally occurring elements to 98.
What is atomic number?
The atomic number of an element is equal to the total number of protons in the nucleus of the atoms of that element. The atomic number can provide...
What is the atomic number and mass number?
The number of protons and the number of neutrons shall determine the mass number of an element. Since the isotopes of an element have slightly diff...
Can two different elements have the same atomic number?
Atoms from two different elements may have the same neutron count, but never the same proton count. The number of protons is unique to the element...
How do we calculate atomic mass?
Add the mass of protons and neutrons to compute the atomic mass of a single atom of an element. Example: Find the atomic mass of a carbon isotope w...
Why is atomic number important?
Atomic number is called the number of protons in an atom. This number is very important, because it is unique to a given element’s atoms. An elemen...
What is the atomic number of an element?
The atomic number refers to the number of protons found in the atom of an element . Elements can be categorized into three major groups that include metals, nonmetals, and metalloids. The elements found on the left side of the periodic table are typically metals. While the elements on the right side of the periodic table are non-metals.
How many elements are in pure form?
Thirty-two of the 98 elements are in their pure form. The rest exist as compounds. Eighty of the natural elements are stable, meaning that they cannot be subjected to radioactive decay. Ten of the 98 elements only exist in trace amounts. Typically, all the elements of the periodic table with a higher atomic number than lead are unstable, ...
What is the difference between francium and native elements?
Native elements, on the other hand, are naturally occurring elements in an uncombined form.
What does the number of protons in an element mean?
The number of protons in an element gives the atomic number of the element. An element refers to a substance made of atoms of the same kind. All the atoms in a particular element bear the same atomic number. Elements cannot be broken further into smaller substances using chemical reactions. However, they can only be transformed into other elements ...
What are the properties of an element?
The periodic table outlines each element’s electron configuration, the atomic number of the element, and the chemical properties of the element. The atomic number refers to the number of protons found in the atom of an element.
What are the elements that are found naturally?
Non-metals that fall into this category include nitrogen, oxygen, and carbon.
Is synthetic element unstable?
However, unlike natural elements that can be handled, these synthetic elements are likely to be unstable, thus decaying quickly. Nonetheless, there is a possibility for more exciting discoveries in the atomic world.
How many elements are in the periodic table?
The table below consists of 118 elements of the periodic table, sorted by atomic number, atomic weight, symbols, density, discovered year and the group.
What is the atomic number of an element?
The atomic number of an element is equal to the total number of protons in the nucleus of the atoms of that element. The atomic number can provide insight into the electronic configuration of the element. For example, carbon has an electron configuration of [He] 2s 2 2p 2, since its atomic number is 6.
What is the number of protons in the nucleus called?
The number of protons in the nucleus is called the atomic number. The atomic number of each element is unique.
Why is the atomic number of each element unique?
While the atomic number always stays the same some elements have atoms with different atomic mass numbers. This is because some elements have a different number of neutrons in the nucleus.
How to find the mass of an element?
The number of protons and the number of neutrons shall determine the mass number of an element. Since the isotopes of an element have slightly different mass numbers, it calculates the atomic mass by obtaining the mean of the mass numbers for its isotopes.
How can periodic trends be observed?
Periodic trends in the properties of the elements can be observed down the groups and across the periods of the modern periodic table. Every chemical element has a specific atomic number, which provides insight into the number of protons present within its nucleus.
Why is the atomic number important?
This number is very important, because it is unique to a given element’s atoms. An element’s atoms all have the same number of protons and each element has a different number of protons in its atoms. Test your knowledge on periodic table elements.
Is element 140 possible?
The current periodic table has proven up for the element with atomic number 118. As of 2020, no elements with an atomic number greater than 118 has successfully incorporated. … However, in real-life science, element 140 has yet to be identified.
Is there a 120th element?
Density (near r.t. ) Unbinilium, also known as eka-radium or simply element 120, is the hypothetical chemical element in the periodic table with symbol Ubn and atomic number 120. Unbinilium has not yet been synthesized, despite multiple attempts from German and Russian teams. …
What is element 140?
Corbomite (symbol Ct) is a chemical element, atomic number 140 on the periodic table.
Is element 119 a metal?
Element 119 is expected to be a typical alkali metal with a +1 oxidation state.
What is the most rare element?
Leimbach et al.) A team of researchers using the ISOLDE nuclear-physics facility at CERN has measured for the first time the so-called electron affinity of the chemical element astatine, the rarest naturally occurring element on Earth.
What is the hardest element?
Chances are you have seen it. The hardest pure element is carbon in the form of a diamond. Diamond is not the hardest substance known to man. Some ceramics are harder, but they consist of multiple elements.
Who is the smallest element?
The element which has the smallest atomic mass is Hydrogen (H), which has a proton and an electron. The atomic radius decreases as we move from left to right along the period. And The atomic radius increases down the group. Thus, helium is the smallest element, and francium is the largest.
What is the number of elements naturally occurring?
However, it turns out there are other elements that occur in trace amounts naturally. This brings the number of naturally occurring elements to 98.
How many elements are there in nature?
The first 91 elements occur in nature, plus a few others, bringing the total to 98 natural elements. Digital Art/Getty Images. Dr. Helmenstine holds a Ph.D. in biomedical sciences and is a science writer, educator, and consultant. She has taught science courses at the high school, college, and graduate levels.
What are rare elements?
The rare elements are produced by radioactive decay and other nuclear processes of more common elements. For example, francium is found in pitchblende as the result of alpha decay of actinium.
What are elements that are not native to nature?
While many elements occur in nature, they might not occur in pure or native form. There are only a few native elements. These include the noble gases, which don't readily form compounds, so they are pure elements. Some of the metals occur in native form, including gold, silver, and copper. Nonmetals including carbon, nitrogen, and oxygen occur in native form. Elements that occur naturally, yet not in native form, include the alkali metals, alkaline earth, and rare earth elements. These elements are found bound in chemical compounds, not in pure form.
Where is technetium 99 found?
Minute amounts of technetium-99 have been found in uranium-rich pitchblende. Elements 93–98 ( neptunium, plutonium, americium, curium, berkelium, and californium) were all first artificially synthesized and isolated in the Lawrence Berkeley National Laboratory at the University of California, Berkeley.
Which elements are found naturally but not in native form?
Elements that occur naturally, yet not in native form, include the alkali metals, alkaline earth, and rare earth elements. These elements are found bound in chemical compounds, not in pure form. Cite this Article. Format.
Is Technetium a stable isotope?
Technetium is one of the newer elements added to the list. Technetium is an element with no stable isotopes. It is produced artificially by bombarding samples of molybdenum with neutrons for commercial and scientific uses and was widely believed to be nonexistent in nature. This has turned out to be untrue.
How many elements are in the periodic table?
Based on an earlier (1882) model of T. Bayley, J. Thomsen in 1895 devised a new table. This was interpreted in terms of the electronic structure of atoms by Niels Bohr in 1922. In this table there are periods of increasing length between the noble gases; the table thus contains a period of 2 elements, two of 8 elements, two of 18 elements, one of 32 elements, and an incomplete period. The elements in each period may be connected by tie lines with one or more elements in the following period. The principal disadvantage of this table is the large space required by the period of 32 elements and the difficulty of tracing a sequence of closely similar elements. A useful compromise is to compress the period of 32 elements into 18 spaces by listing the 14 lanthanoids (also called lanthanides) and the 14 actinoids (also called actinides) in a special double row below the other periods.
What is the long period form of the periodic system?
Long-period form of periodic system of elements. Encyclopædia Britannica, Inc. With the discovery of the noble gases helium, neon, argon, krypton, radon, and xenon by Lord Rayleigh (John William Strutt) and Sir William Ramsay in 1894 and the following years, Mendeleyev and others proposed that a new “zero” group to accommodate them be added to ...
What elements did Mendeleyev predict?
Mendeleyev was also able to predict the existence, and many of the properties, of the then undiscovered elements eka-boron, eka-aluminum, and eka-silicon, now identified with the elements scandium, gallium, and germanium, respectively. Similarly, after the discovery of helium and argon, the periodic law permitted the prediction of the existence ...
How does the atomic weight of an element show its position in the periodic system?
That the exact atomic weight of an element is of small significance for its position in the periodic system is shown by the existence of isotopes of every element —atoms with the same atomic number but different atomic weights. The chemical properties of the isotopes of an element are essentially the same, and all the isotopes of an element occupy the same place in the periodic system in spite of their differences in atomic weight.
Which elements were put in positions out of the order of atomic weights?
In the pairs argon and potassium, cobalt and nickel, and tellurium and iodine, for example, the first element had the greater atomic weight but the earlier position in the periodic system. The solution to this difficulty was found only when the structure of the atom was better understood.
How many spaces are there in a period of 32?
A useful compromise is to compress the period of 32 elements into 18 spaces by listing the 14 lanthanoids (also called lanthanides) and the 14 actinoids (also called actinides) in a special double row below the other periods.
What is the short period of the periodic table?
The “short-period” form of the periodic table, with Groups 0, I , II,…VIII, became popular and remained in general use until about 1930. Short-period form of periodic system of elements, listing the elements known by 1930. At that time it was not clear that thorium (90), protactinium (91), and uranium (92) were part of the actinide series, ...
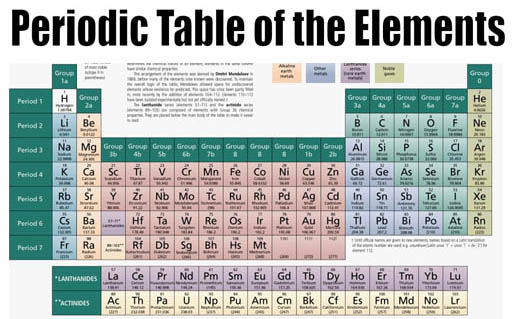
Summary
Classification of elements
Many terms have been used in the literature to describe sets of elements that behave similarly. The group names alkali metal, alkaline earth metal, pnictogen, chalcogen, halogen, and noble gas are acknowledged by IUPAC; the other groups can be referred to by their number, or by their first element (e.g., group 6 is the chromium group). Some divide the p-block elements from groups 13 to …
Overview
The periodic table is a 2-dimensional structured table. The elements are placed in table cells, in reading order of ascending atomic number. The table columns are called groups, the rows are called periods. The breaks at the end of each period occur according to a repetition (or periodicity) of physical and chemical properties of the elements.
Periodic trends
As chemical reactions involve the valence electrons, elements with similar outer electron configurations may be expected to react similarly and form compounds with similar proportions of elements in them. Such elements are placed in the same group, and thus there tend to be clear similarities and trends in chemical behaviour as one proceeds down a group. As analogous configurations return …
History
In 1817, German physicist Johann Wolfgang Döbereiner began to formulate one of the earliest attempts to classify the elements. In 1829, he found that he could form some of the elements into groups of three, with the members of each group having related properties. He termed these groups triads. Chlorine, bromine, and iodine formed a triad; as did calcium, strontium, and barium; lithi…
Current questions
Although the modern periodic table is standard today, some variation can be found in period 1 and group 3. Discussion is ongoing about the placements of the relevant elements. The controversy has to do with conflicting understandings of whether chemical or electronic properties should primarily decide periodic table placement, and conflicting views of how the evidence should be used. A similar potential problem has been raised by theoretical investigations of the superheav…
Future extension beyond the seventh period
The most recently named elements – nihonium (113), moscovium (115), tennessine (117), and oganesson (118) – completed the seventh row of the periodic table. Future elements would have to begin an eighth row. These elements may be referred to either by their atomic numbers (e.g. "element 119"), or by the IUPAC systematic element names which directly relate to the atomic …
Alternative periodic tables
The periodic law may be represented in multiple ways, of which the standard periodic table is only one. Within 100 years of the appearance of Mendeleev's table in 1869, Edward G. Mazurs had collected an estimated 700 different published versions of the periodic table. Many forms retain the rectangular structure, including Janet's left-step periodic table (pictured below), and the m…