What is the most common metal on the periodic table?
What are the most common elements on the periodic table?
- Hydrogen (H) Hydrogen is the most abundant element in the universe, and as its atomic number suggests, it’s pretty important.
- Lithium (Li) Lithium is the MVP of modern technology.
- Beryllium (Be)
- Boron (B)
- Carbon (C)
- Nitrogen (N)
- Oxygen (O)
- Fluorine (F)
What is the percentage of metals in the periodic table?
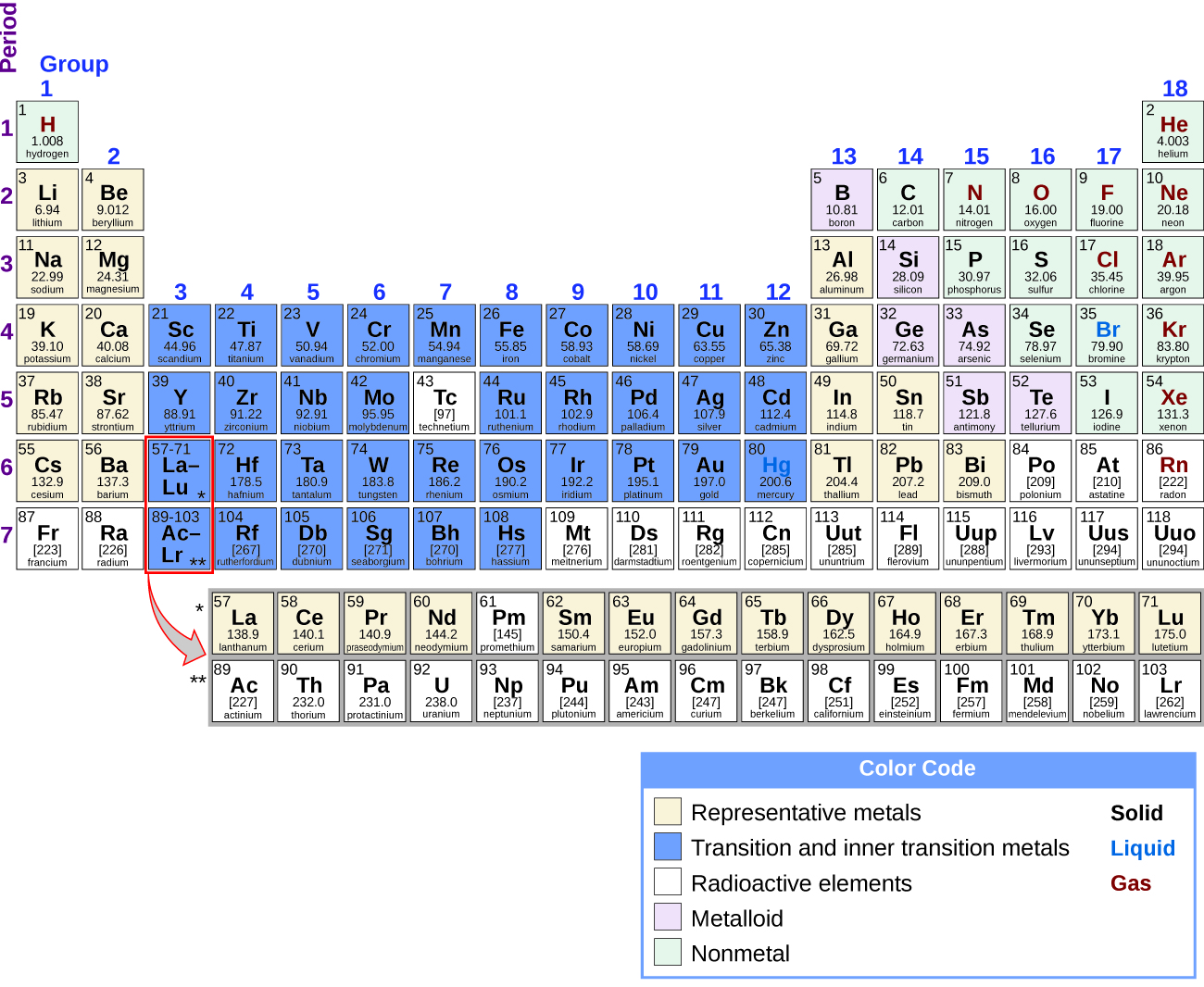
About what percentage of elements in the Periodic Table are Metals? (A) 45%. (B) 55%. (C) 65%. (D) 75%. The answer is: (D) 75%. It depends on the definition of the metals because of the slight differences in metals, nonmetals, and metalloids. However, around 75% of elements in the periodic table are metals.
What are the main metals in the periodic table?
The six alkaline earth metals are:
- beryllium
- magnesium
- calcium
- strontium
- barium
- radium
How are metals classified in the periodic table?
Types of Metals in the Periodic Table
- Alkali Metals. Alkali metals are present at the top left of the periodic table. ...
- Alkaline Earth Metals. Alkaline earth metals are also present at the top left of the periodic table. ...
- Transition Metals. Transition metals are present in the middle of the periodic table. ...
- Lanthanides. Lanthanides are also found in the middle of the periodic table. ...
- Actinides. ...

How many electrons are in an alkaline metal?
All the alkaline earth metals have 2 electrons in their outermost orbit.
How many rare earth metals are there?
There are total 17 Rare Earth metals on the Periodic table. Rare Earth Metals includes all the 15 Lanthanides as well as scandium (Sc) and yttrium (Y). So total 15 + 2 = 17 Rare Earth metals.
What is the inner transition metal?
The answer is: These elements have somewhat similar properties like that of transition metals, plus they are the elements of group 3 only, but they are placed at the bottom of the Periodic table as the inner section of group 3. Hence they are known as inner transition metals.
Where are alkaline earth metals located?
Alkaline earth metals are located on the left side of the Periodic table in group 2. Alkaline earth metals are also the reactive type of metals but they are less reactive as compared to alkali metals. All the alkaline earth metals have 2 electrons in their outermost orbit.
Which metal is the most reactive?
Alkali metals are the most reactive type of metals from the entire Periodic table of elements. As we move down the group from top to bottom in the group 1, the reactivity of alkali metals increases. List of alkali metals with atomic number, symbol and name. Atomic number. Symbol.
Where are transition metals found?
Transition metals are found in the middle part of the Periodic table (from Group 3 to group 11).
What color is heavy metal?
These heavy metals are displayed on the Periodic table with red color (see above image)
Where are metals on the periodic table?
Location of Metals on the Periodic Table. Over 75% of the elements are metals, so they fill most of the periodic table. Metals are on the left side of the table. The two rows of elements below the main body of the table (the lanthanides and actinides) are metals.
What are the elements that make up metals?
The metals consist of the alkali metals, alkaline earths, transition metals, lanthanides, and actinides. Here is a list of metals, their location on the periodic table, their properties, and uses.
What happens to the atoms of metals in a reaction?
Atoms of metals lose electrons in reactions. In other words, they form cations.
What are the characteristics of metals?
Metals have low electronegativities. They are malleable – able to be pounded into sheets. They are ductile – can be pulled into wires. Metals have high density values (exceptions: lithium, potassium, and sodium). Most metals corrode in air or seawater. Atoms of metals lose electrons in reactions.
What are metals used for?
Metals find use in every aspect of life. Here is a list of some of their uses: 1 Structural components 2 Containers 3 Wires and electrical appliances 4 Heat sinks 5 Mirrors 6 Coins 7 Jewelry 8 Weapons 9 Nutrition (iron, copper, cobalt, nickel, zinc, molybdenum)
Is metal a solid?
Metals are solid at room temperature (with the exception of mercury). Metals are shiny, with a metallic luster. Most metals have a high melting point. Most are good conductors of heat. Most are good electrical conductors. They have low ionization energies. Metals have low electronegativities.
What are metals in Periodic table?
Metals are the elements which have the tendency to donate or lose electrons to form positive ions.
Where are metals located on the periodic table?
The metals are located on the left side of the Periodic Table.
How many electrons do metals lose in a chemical reaction?
The atoms or metals have generally 1, 2 or 3 electrons in the outermost orbit, and they lose these electrons during a chemical reaction.
How many rare earth metals are there?
There are total 17 Rare Earth metals on the Periodic table. Rare Earth Metals includes all the 15 Lanthanides as well as scandium (Sc) and yttrium (Y). So total 15 + 2 = 17 Rare Earth metals.
Which group of metals is the most reactive?
They are the Alkali metals of group 1. In 1st group, as we move down from top to bottom, the reactive of metals increases. Thus the bottom most element of group 1 (i.e francium) is the most reactive metal on the Periodic table. ( Note: Francium is a laboratory made element.
Why do metals make a ringing sound?
Metals produce ringing sound when they are stuck hard. This indicates that metals are sonorous in nature.
What are the elements in group 3 to group 12?
The elements lying in group 3 to group 12 are known as Transition metals (or transition elements). Transition metals form a bridge between the chemically active metals of s-block elements and the less active elements of Groups 13 and 14. Thus these metals are known as “Transition metals”.
Who created the periodic table?
The creator of the periodic table, Dmitri Mendeleev, in 1869 began collecting and sorting known properties of elements, like he was playing a game, while traveling by train.
Can periodic table games be used for grade?
The periodic table game available on this page is for entertainment purposes only, and should not be used to grade students on their knowledge of chemical elements.
Does the Modern Periodic Table Change? If So, How and Who Does That?
The periodic table as we know it today is managed by the International Union of Pure and Applied Chemistry, or IUPAC (eye-you-pack).
How many categories are there in the periodic table?
The elements of the periodic table shown here are divided into nine categories; six for the metals, and two for nonmetals, and a metalloid category. The nine categories (or sets) correspond to those found in the literature for the applicable part of the periodic table. Different authors may use different categorisation schema depending on the properties of interest.
What is the periodic table?
The periodic table, also known as the periodic table of elements, is a tabular display of the chemical elements, which are arranged by atomic number, electron configuration, and recurring chemical properties. The structure of the table shows periodic trends. The seven rows of the table, called periods, generally have metals on ...
What is the atomic number plotted against?
Atomic number plotted against atomic radius, excluding the noble gases. Atomic radii vary in a predictable and explainable manner across the periodic table. For instance, the radii generally decrease along each period of the table, from the alkali metals to the noble gases; and increase down each group.
What are metals and nonmetals?
In chronological order, this section discusses metals and nonmetals (and metalloids); categories of elements; groups and periods; and periodic table blocks. While the recognition of metals as solid, fusible and generally malleable substances dates from antiquity, Antoine Lavoisier may have the first to formally distinguish between metals and nonmetals ('non-métalliques') in 1789 with the publication of his 'revolutionary' Elementary Treatise on Chemistry. In 1811, Berzelius referred to nonmetallic elements as metalloids, in reference to their ability to form oxyanions. In 1825, in a revised German edition of his Textbook of Chemistry, he subdivided the metalloids into three classes. These were: constantly gaseous 'gazolyta' (hydrogen, nitrogen, oxygen); real metalloids (sulfur, phosphorus, carbon, boron, silicon); and salt-forming 'halogenia' (fluorine, chlorine, bromine, iodine). Only recently, since the mid-20th century, has the term metalloid been widely used to refer to elements with intermediate or borderline properties between metals and nonmetals. Mendeleev published his periodic table in 1869, along with references to groups of families of elements, and rows or periods of his periodic table. At the same time, Hinrichs wrote that simple lines could be drawn on a periodic table in order to delimit properties of interest, such as elements having metallic lustre (in contrast to those not having such lustre). Charles Janet, in 1928, appears to have been the first to refer to the periodic table's blocks.
How many electrons are in neon?
The electron configuration for neon, for example, is 1s 2 2s 2 2p 6. With an atomic number of ten, neon has two electrons in the first shell, and eight electrons in the second shell; there are two electrons in the s subshell and six in the p subshell. In periodic table terms, the first time an electron occupies a new shell corresponds to ...
What are the columns of periodic table called?
The seven rows of the table, called periods, generally have metals on the left and nonmetals on the right. The columns, called groups , contain elements with similar chemical behaviours.
When is the 150th anniversary of the periodic table?
In celebration of the periodic table's 150th anniversary, the United Nations declared the year 2019 as the International Year of the Periodic Table, celebrating "one of the most significant achievements in science".
How many elements are in the periodic table?
The table below consists of 118 elements of the periodic table, sorted by atomic number, atomic weight, symbols, density, discovered year and the group.
What is the atomic number of an element?
The atomic number of an element is equal to the total number of protons in the nucleus of the atoms of that element. The atomic number can provide insight into the electronic configuration of the element. For example, carbon has an electron configuration of [He] 2s 2 2p 2, since its atomic number is 6.
What is the number of protons in the nucleus called?
The number of protons in the nucleus is called the atomic number. The atomic number of each element is unique.
Why is the atomic number of each element unique?
While the atomic number always stays the same some elements have atoms with different atomic mass numbers. This is because some elements have a different number of neutrons in the nucleus.
How to find the mass of an element?
The number of protons and the number of neutrons shall determine the mass number of an element. Since the isotopes of an element have slightly different mass numbers, it calculates the atomic mass by obtaining the mean of the mass numbers for its isotopes.
How can periodic trends be observed?
Periodic trends in the properties of the elements can be observed down the groups and across the periods of the modern periodic table. Every chemical element has a specific atomic number, which provides insight into the number of protons present within its nucleus.
Why is the atomic number important?
This number is very important, because it is unique to a given element’s atoms. An element’s atoms all have the same number of protons and each element has a different number of protons in its atoms. Test your knowledge on periodic table elements.