/Periodic-Table-Metals-56a12db33df78cf772682c44.png)
Where are nonmetals on the periodic table?
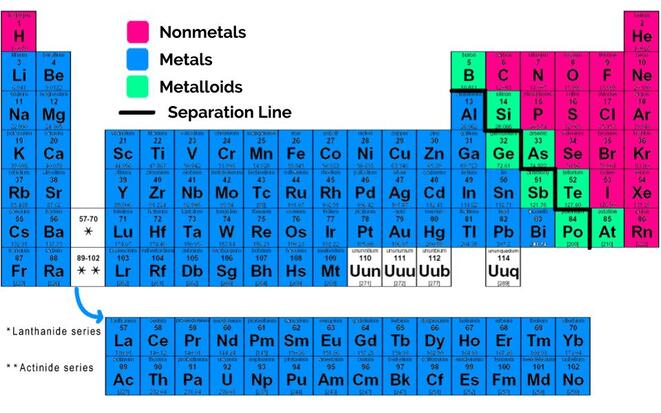
Updated February 25, 2020. The nonmetals or non-metals are a group of elements located on the right side of the periodic table ( except for hydrogen, which is on the top left). These elements are distinctive in that they typically have low melting and boiling points, don't conduct heat or electricity very well, and tend to have high ionization ...
How many elements are in the nonmetal group?
There are 7 elements that belong to the nonmetals group: Although these are the elements in the group nonmetals, there are two additional element groups that could be included, since the halogens and noble gases also are types of nonmetals.
What are the oxidation numbers of nonmetals?
Atoms of these elements have oxidation numbers of +/- 4, -3, and -2.
Is carbon a nonmetal?
Nonmetals are classified based on their properties under ordinary conditions. Metallic character isn't an all-or-nothing property. Carbon, for example, has allotropes that behave more like metals than nonmetals. Sometimes this element is considered to be a metalloid rather than a nonmetal.
What are the characteristics of nonmetals?
Nonmetals exhibit very different properties from metals. Examples of nonmetals include oxygen, chlorine, and argon. Nonmetals display some or all of the following characteristics: 1 Dull appearance 2 Usually brittle 3 Poor conductors of heat and electricity 4 Usually less dense, compared to metals 5 Usually low melting point of solids, compared with metals 6 Tend to gain electrons in chemical reactions
What are the elements in the periodic table?
Elements of the periodic table are grouped as metals, metalloids or semimetals, and nonmetals. The metalloids separate the metals and nonmetals on a periodic table. Also, many periodic tables have a stair-step line on the table identifying the element groups.
Which element is considered a metal?
Elements to the left of the line are considered metals. Elements just to the right of the line exhibit properties of both metals and nonmetals and are termed metalloids or semimetals. Elements to the far right of the periodic table are nonmetals. The exception is hydrogen (H), the first element on the periodic table.
What are the properties of metals?
Metals exhibit the following properties: Usually solid at room temperature (mercury is an exception) High luster (shiny) Metallic appearance. Good conductors of heat and electricity. Malleable (can be bent and pounded into thin sheets) Ductile (can be drawn into wire) Corrode or oxidize in air and seawater.
What are some examples of metalloids?
Examples of metalloids include boron, silicon, and arsenic. Metalloids have some of the properties of metals and some nonmetallic characteristics. Dull or shiny. Usually conduct heat and electricity, though not as well as metals. Often make good semiconductors.